Clinical Research in Oncology
We participated in the “Clinical Research in Oncology” panel organized by Başkent University as Klinar CRO. ...
17 October, 2025

Local CROs generally employs senior project managers supporting all the CRAs working for the trials as a supervisor with long experience in that area. This helps to focus on the trials with a micromanagement aspect and gives the opportunity to the sponsors to always see a back up person and an experienced controller to help the study to be conducted.
Local CROs are more cost effective including the services fees and the services provided compares with the global CROs. Mainly most of the issues are followed up as a part of project management activities without having separate prices for each and every service item.
With that wide experience in clinical trials in Turkey, local CROs also have an insight of probably issues that can occur in a trial during design, plan and conduct of the trial with pre-planned resolutions for this issues.
Local CROs still serve in our country as a school for clinical trials . Most of the global CROs use the CRAs in only one expertise area instead of training the CRA as a project manager dealing with all activities during a clinical trial makes the integrity of the trial to fail and communication issues generally occur affecting the integrity of the trial.
Local CROs in Turkey have a more expanded database of investigators and sites and they are in touch with many KoLs to collaborate with during the set-up phase of the studies since they have a long-established background in clinical trials in Turkey.
Local CROs are in close relationship with both Ethics Committees and Ministry of Health. The committees and the MoH sometimes can have the opportunity to accelerate the bureaucratic pathways for the local CROs which is an advantage considering the timelines.

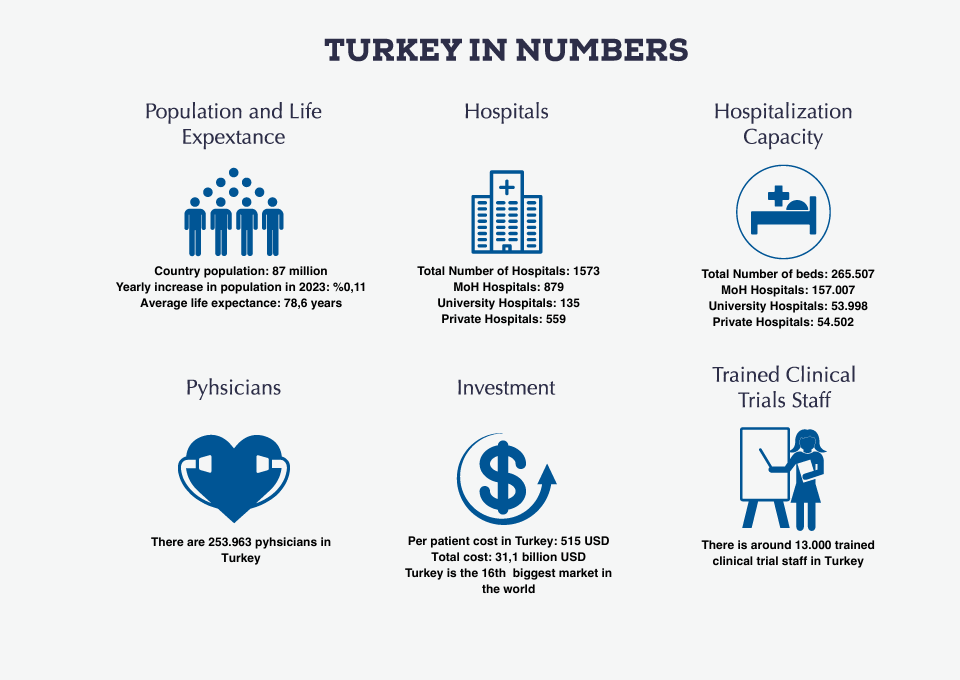
Klinar CRO has a wide range of experience in different therapeutic areas.
Different types of trials such as; Clinical Trials, Post Marketing Trials, Site Management Organization, Electronic Data Capture, ATMP studies, Medical Device Studies and Compassionate Use are being conducted by Klinar CRO.
Clinical Drug Studies
Observation Studies
Medical Devices and Other Studies
Studies
Sites
Investigators
Patients
We participated in the “Clinical Research in Oncology” panel organized by Başkent University as Klinar CRO. ...
17 October, 2025Our general manager Gülden Ortaç will be attending to a webinar on Turkish Regulatory Requirements for Clinical Developm...
08 July, 2025For clear strategies for your project with flexible, professional, proactive teamwork with competitive costs.